PB: Polybutene
- Short Name
- PB
- Name
- Polybutene
- Group
- CTP - Commodity Thermoplastics
- General Properties
- Chemical Formula
- Structural Formula
-
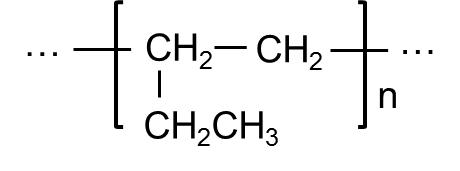
Properties
- Glass Transition Temperature
- -30 to -20 °C
- Melting Temperature
- 115 to 135 °C
- Melting Enthalpy
- 128 J/g
- Decomposition Temperature
- 450 to 460 °C
- Young's Modulus
- 240/600 to 700 MPa
- Coefficient of Linear Thermal Expansion
- 110 to 140 *10¯6/K
- Specific Heat Capacity
- 1.8 to 2.0 J/(g*K)
- Thermal Conductivity
- 0.17 to 0.22 W/(m*K)
- Density
- 0.89/0.91 to 0.94 g/cm³
- Morphology
- Semi-crystalline thermoplastic
- General properties
- High-temperature resistance. Good chemical resistance to solvents, oils, fats, acids and bases
- Processing
- Injection moulding, extrusion
- Applications
- Pipelines (e.g., underfloor heating, drinking water pipelines) Food packaging (films, often with PE and PP) Hotmelt adhesives Compounds and masterbatches
Internet Links
NETZSCH Measurements
- Instrument
- DSC 204 F1 Phoenix®
- Sample Mass
- 11.06 mg
- Isothermal Phase
- 8 min
- Heating/Colling Rates
- 10 K/min
- Crucible
- Al, pierced
- Atmosphere
- N2 (40 ml/min)
Evaluation
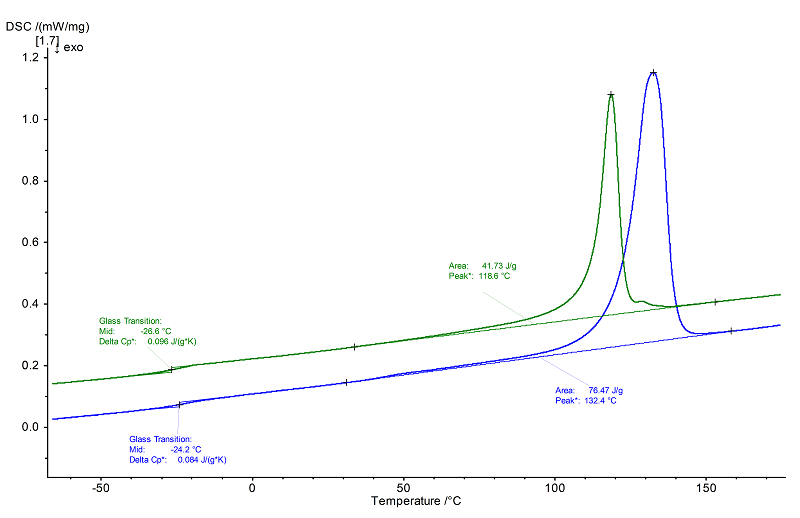
Along with a glass transition at -27°C (midpoint) with a Δcp of approx. 0.10 J/(g·K), the semi-crystalline plastic shows an endothermal melting range – quite narrow for plastics – in the 2nd heating (green) with a main peak temperature of 119°C.
It can be concluded from this peak shape that the molecular weight distribution is largely homogenous. The corresponding heat of fusion amounts to 42 J/g and is therefore significantly smaller than in the 1st heating (blue, 76 J/g). The smaller downstream melting effect at 129°C in the 2nd heating is much closer to the peak temperature of the main melting effect in the 1st heating (133°C). It can therefore be concluded that during the controlled cooling at a cooling rate of 10 K/min, another crystalline phase formed that was not originally present. The glass transition at -24 °C (midpoint) in the 1st heating is similar to that in the 2nd heating, but it shows a bit smaller step height (Δcp) of 0.08 J/(g·K).
It can be concluded from this peak shape that the molecular weight distribution is largely homogenous. The corresponding heat of fusion amounts to 42 J/g and is therefore significantly smaller than in the 1st heating (blue, 76 J/g). The smaller downstream melting effect at 129°C in the 2nd heating is much closer to the peak temperature of the main melting effect in the 1st heating (133°C). It can therefore be concluded that during the controlled cooling at a cooling rate of 10 K/min, another crystalline phase formed that was not originally present. The glass transition at -24 °C (midpoint) in the 1st heating is similar to that in the 2nd heating, but it shows a bit smaller step height (Δcp) of 0.08 J/(g·K).